Life
Scientists have learnt to convert any plastic waste into propane
Environmental pollution by plastic is one of the main environmental problems right now. Its main difficulty lies not even in the fact that not many people hand over plastic items for recycling, but in the fact that until now there has been no perfect technology for this very recycling, and a lot of waste has remained dead weight.
The key problem is that there are many types of plastics that require different chemical processes to turn them into a reusable form. As a result, sorting a huge number of bottles, containers and household items becomes economically unfeasible. And now, scientists at the Massachusetts Institute of Technology have reported that the necessary technology has been found.
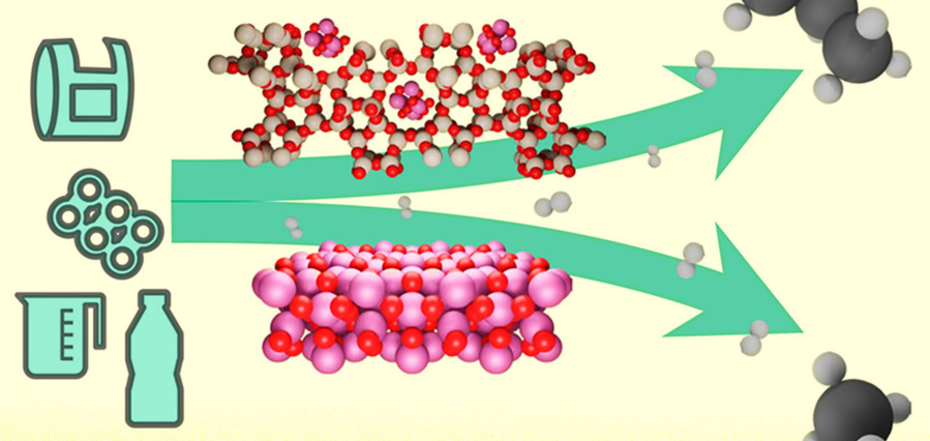
They have developed a chemical process using a metal-based catalyst called cobalt that effectively breaks down various plastics, including the most popular types of polyethylene and polypropylene, into propane, one of the main components of household gas or automotive fuel. In addition, the resulting propane can be used to make new plastics, providing at least a partially closed-loop system for plastic recycling.
The new catalyst easily tackles a key problem in plastic recycling. The long-chain molecules of such materials hold together carbon bonds that are difficult to break to convert plastic into simpler materials suitable for further use. Existing methods of breaking these bonds produced a random mixture of different molecules that required further complex purification and separation into usable compounds. It was impossible to control where the plastic molecule would break.
The catalyst invented by MIT scientists is made of a microporous material called zeolite. It contains cobalt nanoparticles and can selectively destroy various plastic molecules, converting more than 80% of the raw material into propane.
Scientists feared that zeolite would interact poorly with plastic, but it turned out that this material is not only able to pass polymer molecules through itself, but also break them in such a way as to cut off exactly one propane molecule. At the same time, the reaction does not produce unwanted explosive methane. The feedstock can be passed through this catalyst as long as necessary, and it will produce propane again.
The advantage of the method is that the materials needed to recycle plastic - zeolite, cobalt and hydrogen - are fairly cheap and widely available. The catalyst is currently being tested, and engineers are already refining its use to make the method available for industrial-scale plastic recycling. The challenge so far is to determine how much the inks, glues and label materials used in the production of plastic packaging, for example, contaminate the raw materials. And how they can affect the initial reaction product.
As OBOZREVATEL previously reported, the problem of plastic use and the increase in plastic waste has worsened during the coronavirus pandemic. The increase in waste came from hospitals, which needed more masks, gloves, and other protective materials.